Briefing Room
Statement from President Joe Biden on $6.1 Billion in Student Debt Cancellation for 317,000 Borrowers who Attended the Art Institutes
A Proclamation on National Mental Health Awareness Month, 2024
Statement from President Joe Biden on First Quarter 2024 GDP
A Proclamation on Asian American, Native Hawaiian, and Pacific Islanders Heritage Month, 2024
FACT SHEET: Biden-Harris Administration Continues to Call on Congressional Republicans and Internet Service Providers to Keep Americans Connected as the Affordable Connectivity Program Enters Final Month
Remarks by President Biden at the White House Correspondents’ Dinner
FACT SHEET: Biden-Harris Administration Announces $3 Billion to Replace Toxic Lead Pipes and Deliver Clean Drinking Water to Communities Across the Country
Readout of President Joe Biden’s Call with Prime Minister Netanyahu of Israel
FACT SHEET: President Biden Announces up to $6.1 Billion Preliminary Agreement with Micron under the CHIPS and Science Act
Featured Media

Last year, we secured the release of Abigail, a 4-year-old who was being held by Hamas.

Investing in the
American
People
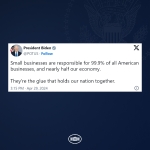
From rebuilding our infrastructure to lowering costs for families, President Biden’s economic agenda is investing in America.
We Want to Hear From You
Send a text message to President Biden, or contact the White House.
Message and data rates may apply.
Reply HELP for help or STOP to cancel.
The
White House
Learn more about the current administration, our country’s former presidents, and the official residence.